One platform to run faster, more inclusive trials, together
Inato unites sponsors, research sites, and patients through one AI-powered platform, expanding access, increasing efficiency, and bringing life-changing treatments to more communities around the world.

Talk to us










Built for global impact. Designed for local access.
Inato is the only platform that brings sponsors and sites together to unlock access to clinical trials, faster and more equitably.
Compliance you
can count on.


Our global presence
50+
Countries
Sites
25+
Sponsors
Trusted by today’s clinical research leaders
We co-develop our technology with forward-thinking pharma and site leaders to ensure every trial is faster, more efficient, and more inclusive.












One intelligent platform to run better trials
Optimize across your portfolio with insights from sites who can deliver on your trials.
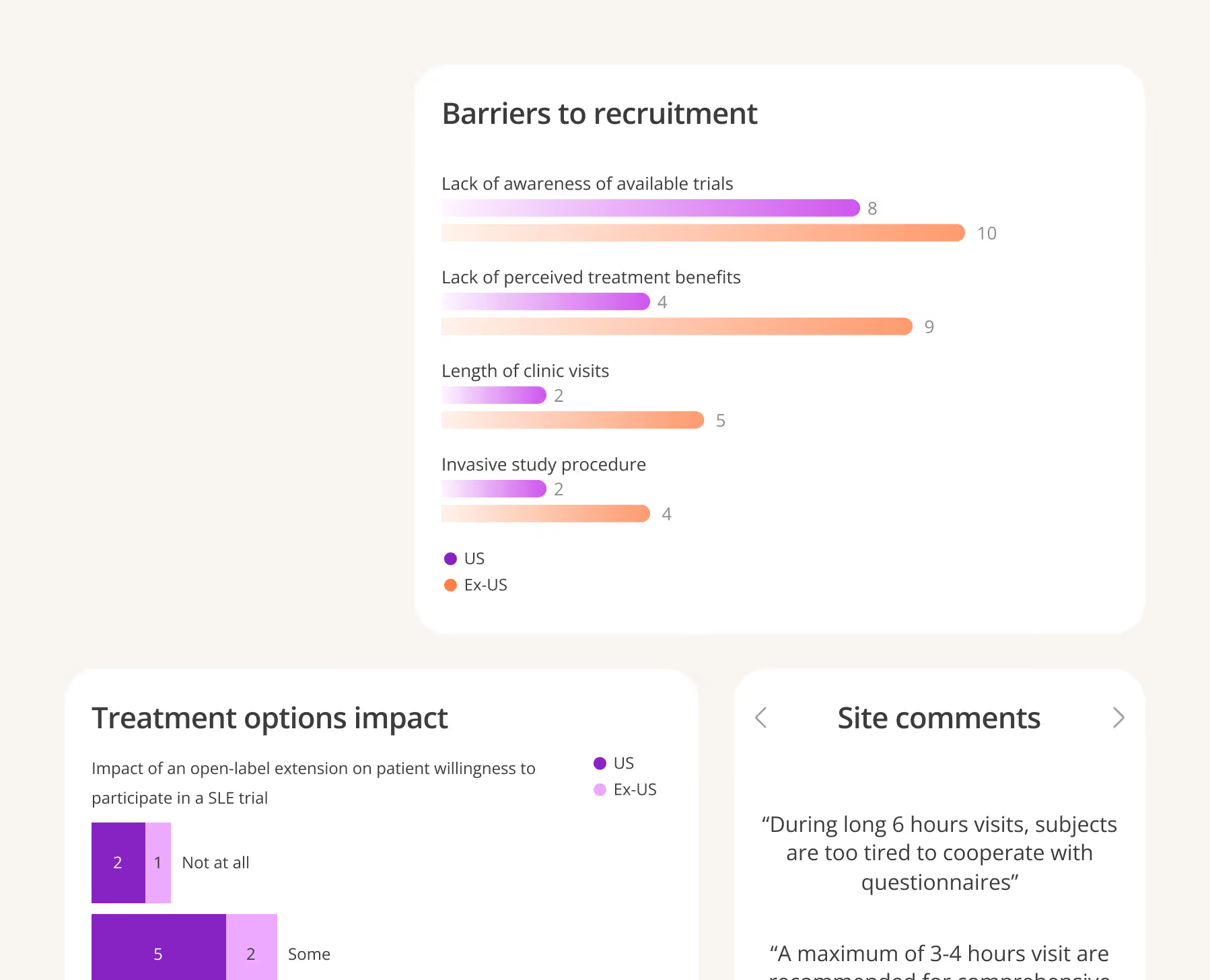
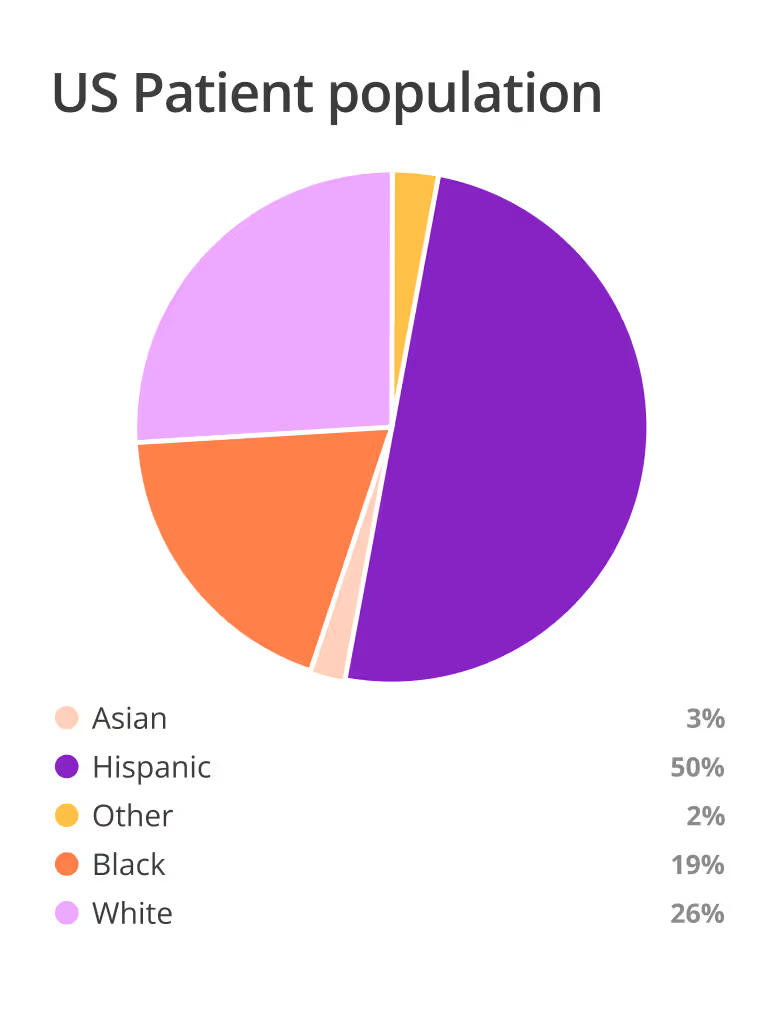
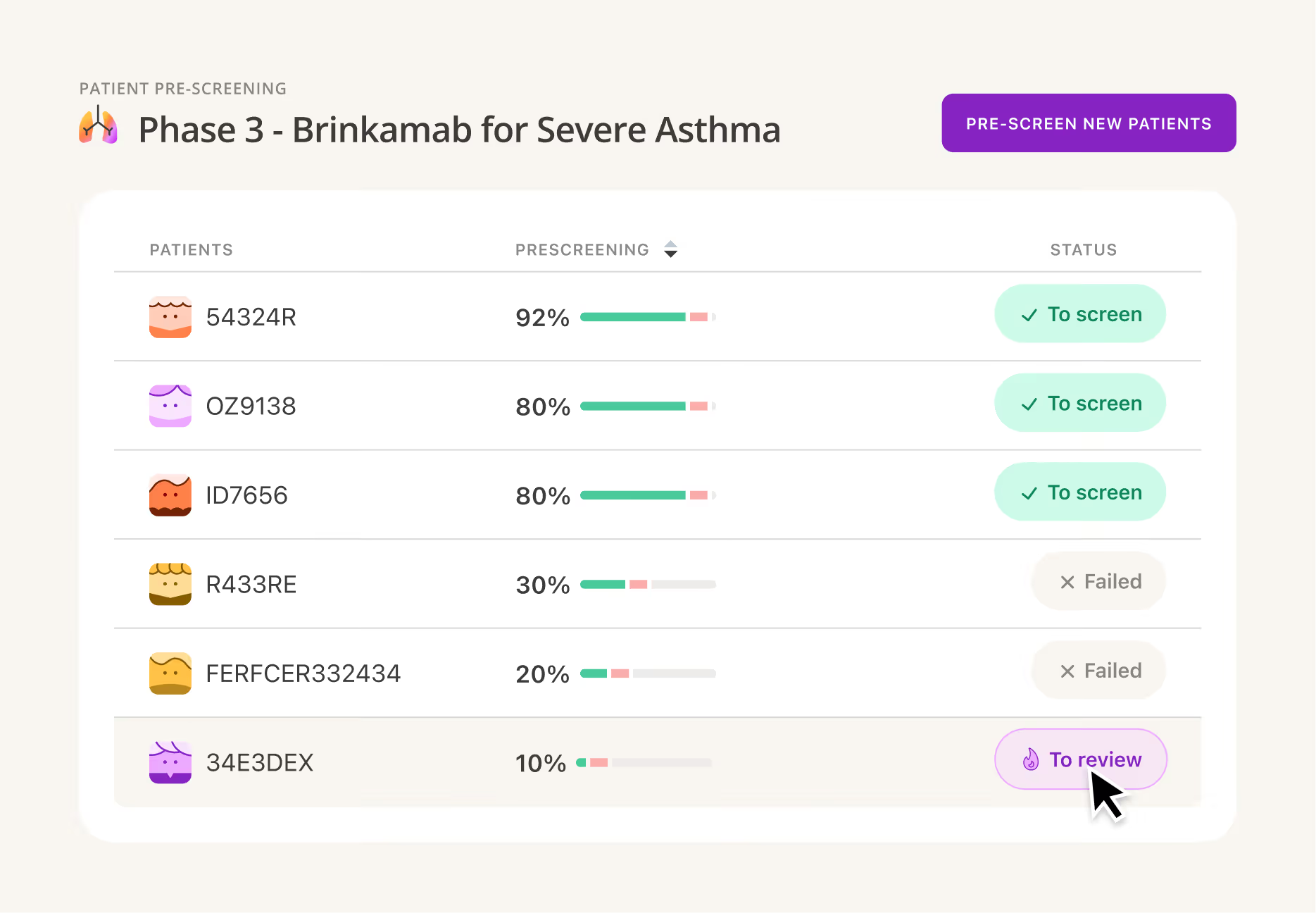
Leverage AI to find the right mix of qualified, motivated sites based on actual capabilities and patient access.
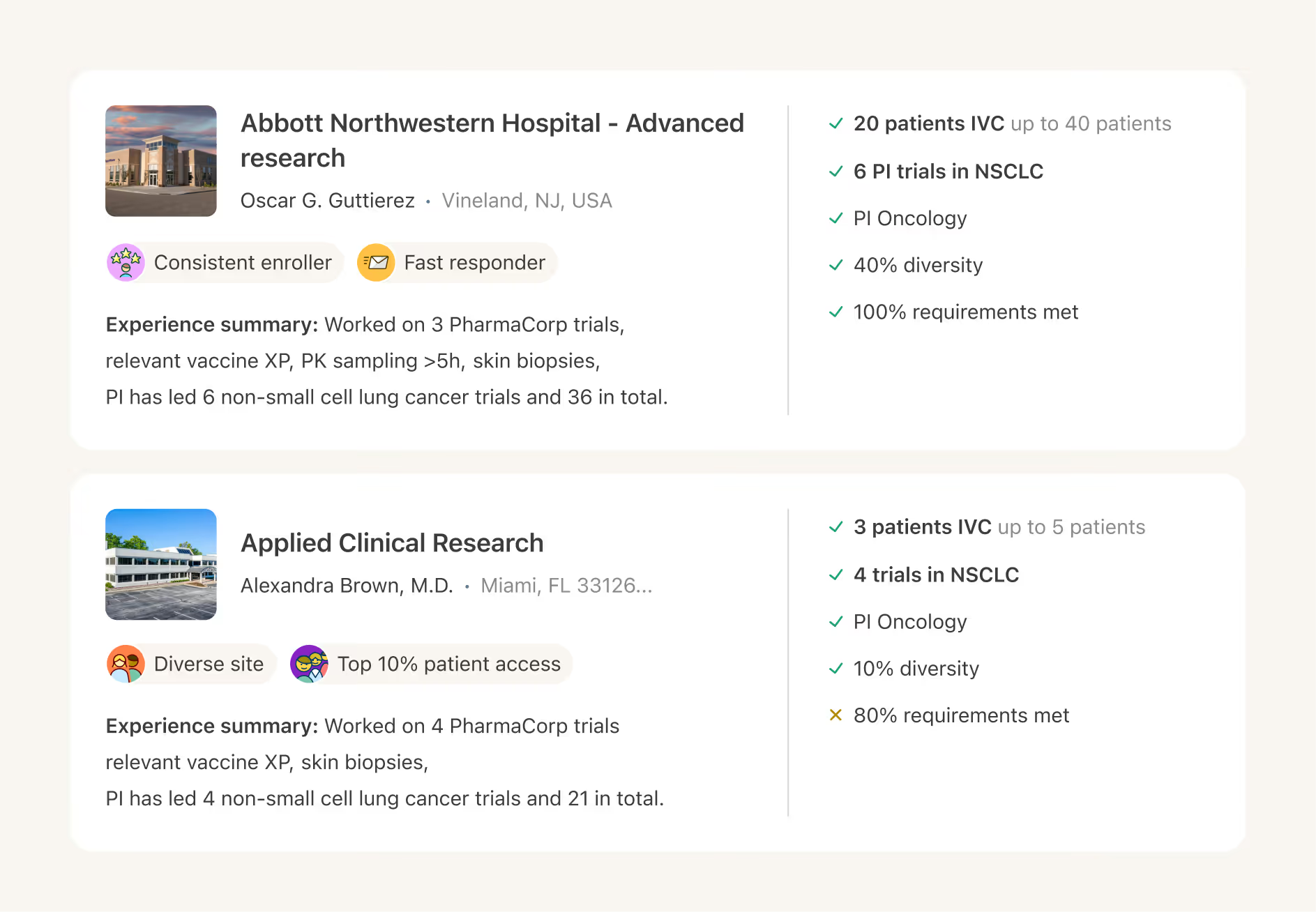
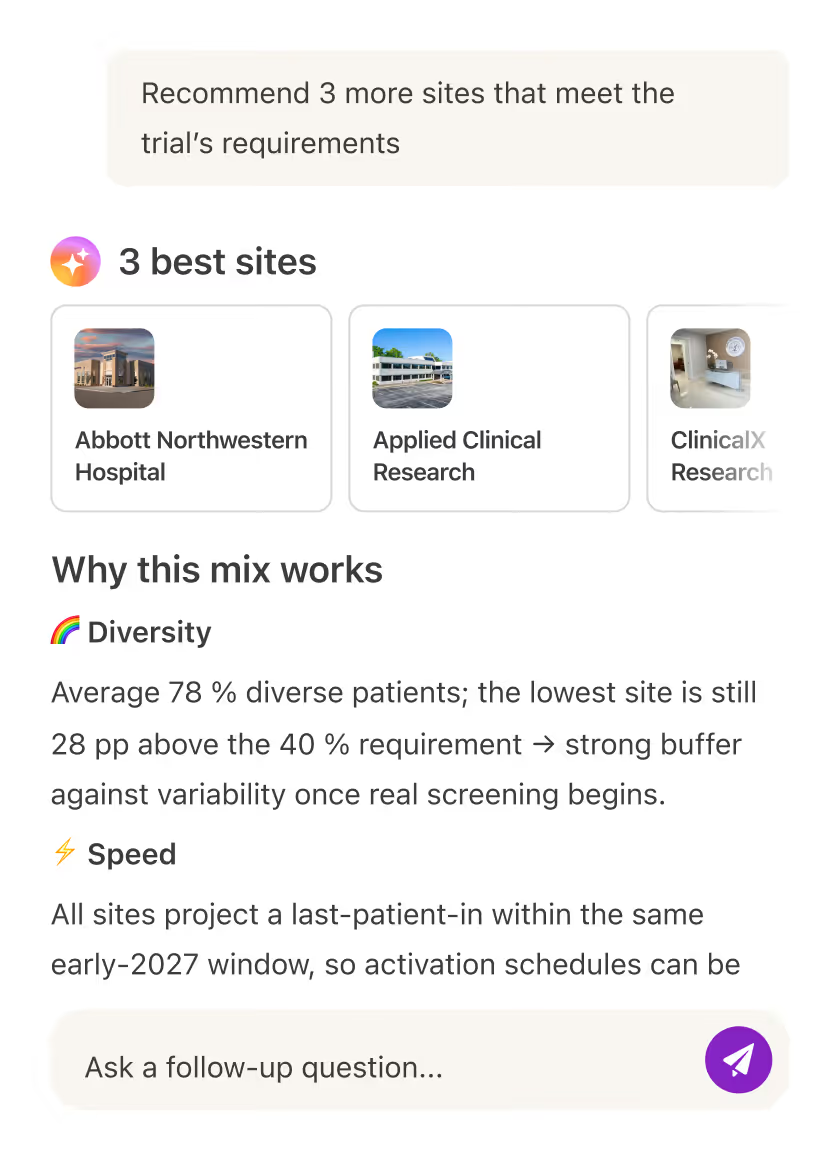
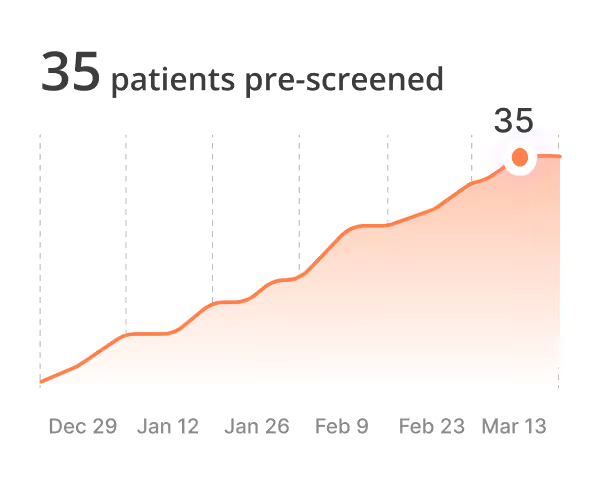
Reduce site burden and accelerate patient enrollment by using AI to automate chart review and pre-screening.